WALS®
Quick Links
WALS® and VectoBac® WDG win Annual Chicago Innovation Award.
Learn more

VectoBac® WDG product information. Learn more
Learn more about how to protect your own district against container mosquitoes.
Get your free WALS for Container Mosquito Management kit. Find publications, research data, application training videos and more supporting use of WALS to control mosquitoes that vector Zika, Dengue, West Nile and Chikungunya viruses.
Learn more
VectoBac WDG is the first bacterial larvicide to complete the World Health Organization Pesticide Evaluation Scheme.
Read the report
Contact a Valent BioSciences public health representative to learn more about our WALS℠ program scientific consulting/analysis and reporting services. Contact us
WALS® and VectoBac® WDG win Annual Chicago Innovation Award.
Learn more

VectoBac® WDG product information. Learn more
Learn more about how to protect your own district against container mosquitoes.
Get your free WALS for Container Mosquito Management kit. Find publications, research data, application training videos and more supporting use of WALS to control mosquitoes that vector Zika, Dengue, West Nile and Chikungunya viruses.
Learn more
VectoBac WDG is the first bacterial larvicide to complete the World Health Organization Pesticide Evaluation Scheme.
Read the report
Contact a Valent BioSciences public health representative to learn more about our WALS℠ program scientific consulting/analysis and reporting services. Contact us
WALS® - More than a product – a layered strategy
WALS® is not merely a product, it’s a layered approach that utilizes cutting-edge formulation technology and a special application system to deliver the highly effective and target-specific biorational larvicide VectoBac® WDG (Bti strain AM65‐52). Building upon more than 20 years of research, Valent BioSciences in partnership with other public health organizations have developed the most target-specific biorational solution for controlling mosquito larvae in difficult-to-find and/or access habitats. And with the increasing potential of vector-borne disease outbreaks from container mosquitoes threatening larger segments of our population, there’s never been a greater need for WALS to help public health professionals meet their Integrated Vector Management (IVM) goals.
The WALS layered approach
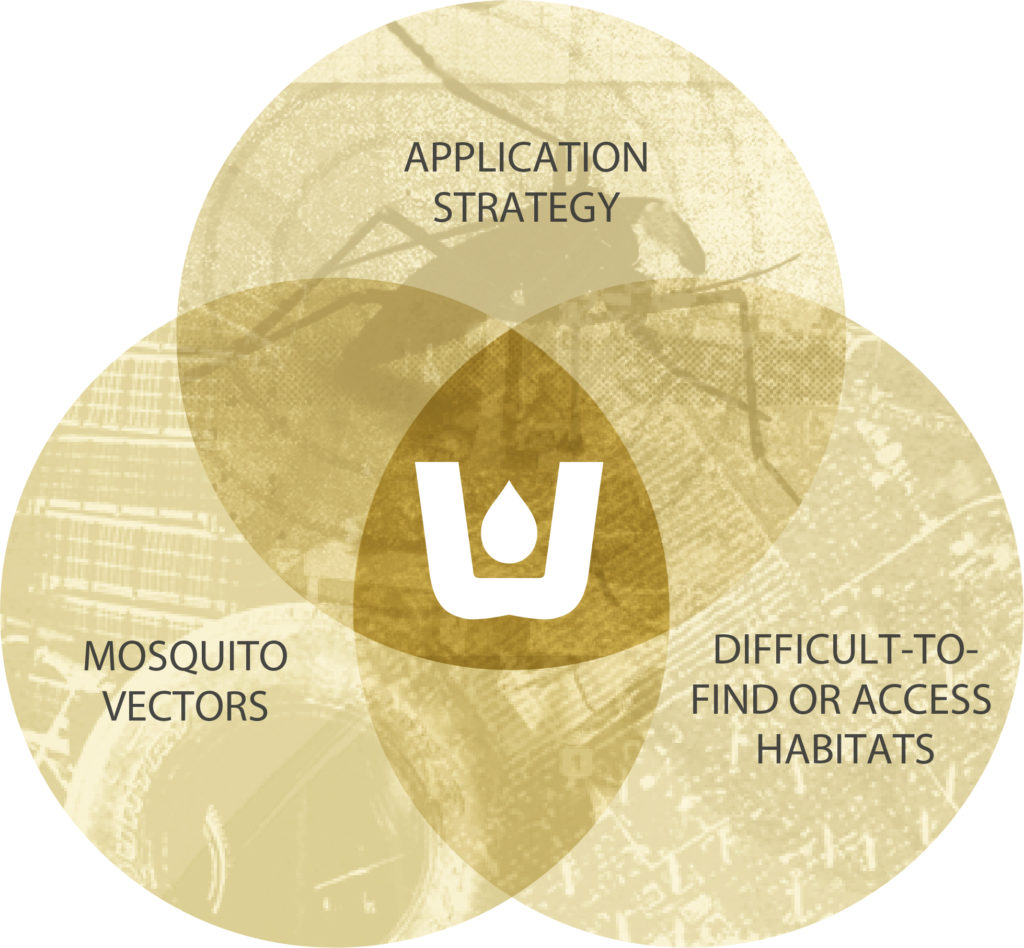
WALS’ solution for the management of vector populations is found at the intersection of three factors: 1) difficult-to-find and/or access larval mosquito habitats, 2) mosquito vectors and 3) application strategy.
Specifically, container mosquito species can vector diseases such as Zika virus, West Nile virus, dengue fever, yellow fever, and chikungunya virus. Larvae of these vector populations often inhabit small/cryptic containers and other difficult-to-find and/or access habitats.
WALS can be applied in a variety of methods for these types of habitats (both artificial and natural containers): Backpack sprayers for targeted coverage of hot spots. Vehicle mounted sprayers for wide-area coverage of specific blocks. And aerial application for rapid coverage of large blocks.
Why it works
Several characteristics uniquely position the WALS strategy to enhance IVM programs aimed at controlling mosquito larvae in these challenging habitats. As a biorational solution, programs can have peace-of-mind that these applications are non-toxic to humans, animals and other non-target organism populations (e.g. beneficial insects), yet meets the efficacy requirements required by public health organizations.
Container mosquitoes, in particular, can breed in a variety of cryptic habitats, from plastic debris spilled in an alley way to the crown of a palm tree. Taking the WALS approach allows one to fully cover this “three-dimensional” target habitat space, reaching larvae wherever they’re growing. The WALS approach allows for a unique biorational droplet size that is capable of moving through space but that will eventually find and drop into small cryptic containers.
Proven to save lives

20+ academic studies

10+ publications

11 countries
Different WALS application methods

Backpack sprayers provide targeted coverage of hot spots.

Aerial application is recommended for rapid coverage of large blocks.

Vehicle-mounted sprayers are used for wide area coverage of specific blocks.
Container mosquitoes
Container mosquitoes – which include such species as Aedes aegypti, Aedes albopictus, and Culex quinquefasciatus/pipiens – can be highly adaptive, aggressive, and opportunistic. They thrive in urban areas where numerous, small and cryptic habitats are plentiful.

Aedes aegypti

Aedes albopictus

Aedes triseriatus

Aedes sierrensis

Culex pipiens
Aedes notoscriptus

Culex quinquefasciatus
Culiseta incidens
Aedes japonicus

Aedes atropalpus
Mode of application
WALS was developed in direct response to the challenges posed by the diversity and sheer number of container mosquito habitats. To effectively control container mosquitoes, operators must deliver an effective, biorational active ingredient into numerous tiny and hard‐to‐reach areas, many not visible to, or accessible by surveillance teams. The WALS approach requires products with unique biorational formulation properties and targeted droplet size ranges to ensure that field technicians are able to reach container mosquito larvae over a wide area.
Five qualifiers define WALS’ unique capabilities:
1. Biorational – not harmful to people, animals, non-target insect populations or the environment
2. Efficacy – controls mosquito vector populations at levels acceptable to public health organizations
3. Formulation – technology that can achieve WALS operational parameters and sustainability
4. Droplet size – droplet range that allows both travel through space but also drops down into targeted container habitats; should be in the ‘extremely fine’ to ‘fine’ (30-235 micron) range
5. Coverage/Access – efficiently treat difficult-to-find and/or access habitats across a “3-dimensional space”; ability to reach mosquito larvae in small, cryptic containers
Case Study: IVM with WALS stops Zika

Provisional data as of March 6, 2019, Centers for Disease Control and Prevention
WALS and VectoBac WDG were critical components in Miami-Dade County’s historic achievement of stopping transmission of locally acquired Zika during the Florida 2016 outbreak. Until recently, the Zika virus – a mosquito-borne flavivirus – was considered to be a relatively harmless disease. But Zika underwent mutations that made if far more aggressive and impactful to humans. Symptoms in more recent outbreaks have been reported as being more severe than with previous Zika infections. Zika has been documented to be spread primarily through the bite of an infected Aedes aegypti mosquito, but other routes of infection have been published and include transmission from pregnant woman to her fetus, sexual transmission, or blood transfusions. Only one person in five will show any symptoms – including fever, rash, joint pain, conjunctivitis, muscle pain, or headache. Other potential outcomes are Guillain‐Barré Syndrome or microcephaly in newborn babies. There are no current vaccine treatments for Zika. Vector control remains the only existing intervention today.

Microcephaly
The more aggressive strain of Zika virus can cause neurological disorders among newborns from infected mothers, including microcephaly. Symptoms of this condition can include a partially collapsed or misshapen skull; decreased brain tissue; damage to the back of the eye; joints with limited range of motion; excessive muscle tone, restricting body movement; and hearing loss.
Zika’s global spread
Over seven decades, the Zika virus has spread across 6 continents and 86 countries and territories.
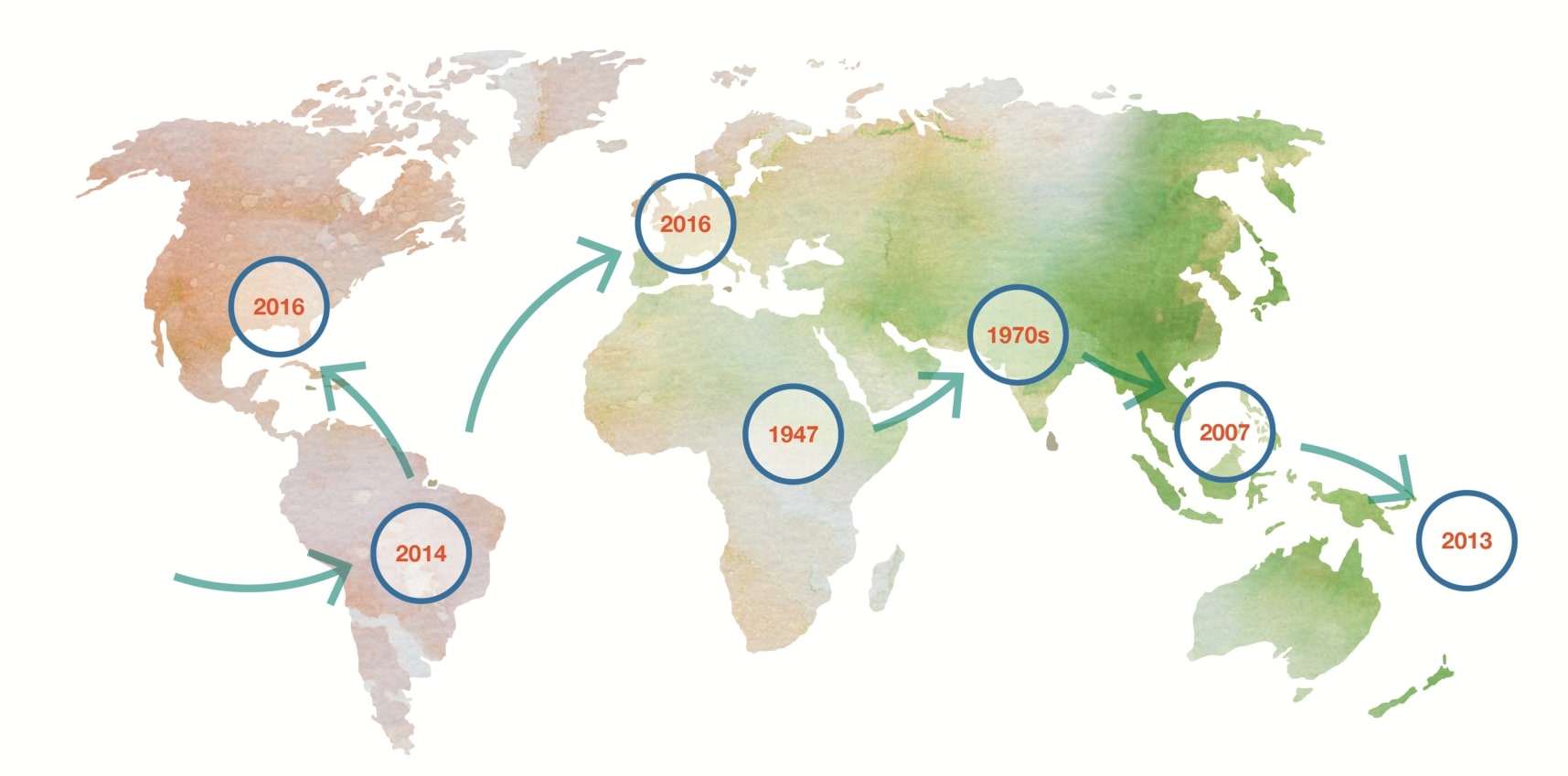
The Zika virus was first identified in Ugandan monkeys in 1947. For the next six decades, it was found in occasional cases of mild human infections across Africa and Asia. In March 2015, Brazil reported a large outbreak of rash illness – soon identified as the Zika virus infection – and within seven months, that country reported an association between Zika and microcephaly. Outbreaks and evidence of transmission soon appeared throughout the Americas, Africa, and other regions globally. To date, a total of 86 countries and territories have reported evidence of the mosquito-transmitted Zika infection.
In late July 2016, the first locally acquired Zika cases in the continental U.S. were confirmed in Miami‐Dade County. By August 31, the Centers for Disease Control (CDC) had confirmed 35 cases of locally acquired Zika in Florida. At that time, adulticides had served as Miami-Dade’s core intervention for mosquito control, but the Zika outbreak prompted the program to expand its IVM approach to include other novel, but proven interventions to help stop transmission. This led to Miami-Dade integrating a WALS strategy (both from aerial and trunk mounted applications) into their program. On August 15, 2016, the Miami Herald was among the first publications to report the dramatic effect of WALS and Vectobac WDG. Janet McCallister, a medical entomologist with the Centers for Disease Control and Prevention, was quoted as saying, “In areas without the larvicide, the adult populations are rebounding much quicker and much higher than in the area [treated] with both.” On September 19, Florida Governor Rick Scott declared Miami‐Dade County Zika-free.
The best tools to protect your district
With the alarming impacts of climate change and dramatic shifts in population growth in urban areas, the spread of infectious diseases are only expected to place greater demands on public health officials globally to manage container mosquitoes.
As a proven IVM strategy, WALS is explicitly designed to help keep the public safe from infectious diseases carried by mosquitoes whose larvae inhabit difficult-to-find and/or access habitats. This approach is a customized blend of two factors that are proven to help better manage these challenging larval habitats: 1) a specially designed application technology joined with 2) a low-risk, target-specific biorational larvicide. Ultimately, WALS is about saving lives by reducing mosquito vector populations to break the disease transmission cycle and reduce the risk of disease.
By incorporating WALS as part of your comprehensive IVM program, you can be fully confident that you’ll have the right tools in place to protect your district from the threat of infectious disease.
Get the WALS for Container Mosquito Management Kit
This is the public health professional’s resource collection to understanding, validating and communicating the effectiveness of WALS programs for container mosquito control.
